Full list of publication: https://www.ncbi.nlm.nih.gov/pubmed/?term=safavi-hemami

Venom Peptide Discovery
Over the past five years we have conducted a world-wide field collection effort to sequence more than 100 different species of venomous marine gastropods by transcriptomics and proteomics (right image: shells of sequenced marine gastropods). By applying several different computational pipelines we have identified more than 40,000 toxin sequences including many entirely new gene families of yet unknown function. This represents one of the largest collection and sequencing effort of a venomous genus to date. We have characterized several of these new toxins and are currently developing bioinformatic tools to identify the most promising future drug leads from these large datasets.
Selected Publications ; Photos of field collections; Contact us if you want to sequence your favorite venomous marine snail!
Venom Insulins – Discovery, Evolution and Biomedical Applications
We discovered that Conus geographus, a cone snail species that releases venom into the water to sedate its prey, uses an unusually small and heavily post-translationally modified insulin, Con-Ins G1, for prey capture. When released into the water, this insulin rapidly induces hypoglycemic shock in fish, the prey of C. geographus. Structure-function analysis of this unique insulin has already provided novel insights into insulin receptor activation and uniquely informed on the design of a fast-acting drug lead for type 1 and late-stage type 2 diabetes. Using our large venom datasets we have since been able to show that venom insulins are widely distributed in cone snail venoms and have identified a panel of additional fast-acting venom insulins that bind to and activate the human insulin receptor. We anticipate that these diverse insulins will serve as unique pharmacological tools for studying insulin receptor activation and isoform distribution and hold tremendous opportunity for current efforts into the design of novel fast-acting therapeutics for diabetes.
Selected publications; Venom insulin structure; CryoEM complex of a venom-human insulin hybrid analog with the human insulin receptor: asymmetric conformation, symmetric conformation; Podcast 2022 Podcast 2019; Selected Podcast 2015

Venom GPCR ligands
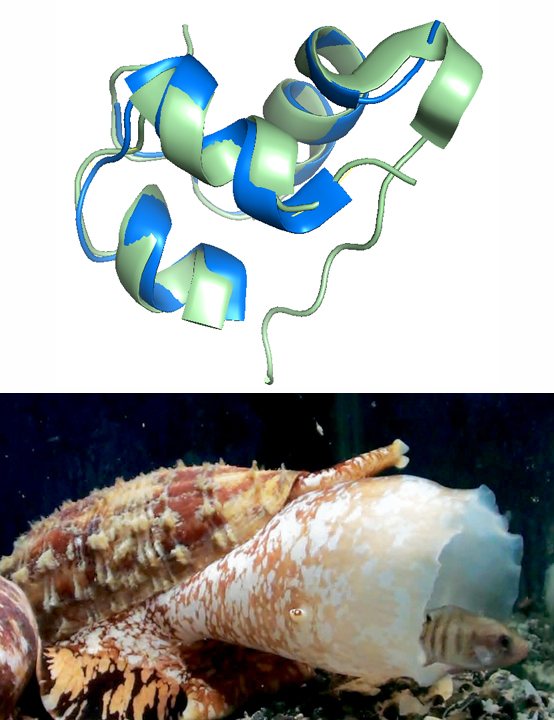
By investigating a deep-water clade of fish-hunting cone snail we recently identified a new family of toxins that mimic somatostatin, a peptide hormone with diverse physiological roles. Remarkably, these venom peptides share structural and functional similarity with somatostatin drug analogs, including a minimized disulfide fold and a D-tryptophan within the disulfide loop. We are currently further investigating these toxins as potential drug leads for pain and inflammation and have identified other GPCR-targeting toxins from venom.
Selected publications; Venom somatostatin (consomatin) structure; Video of cone snail predation strategies; Podcast 2022
Venom evolution
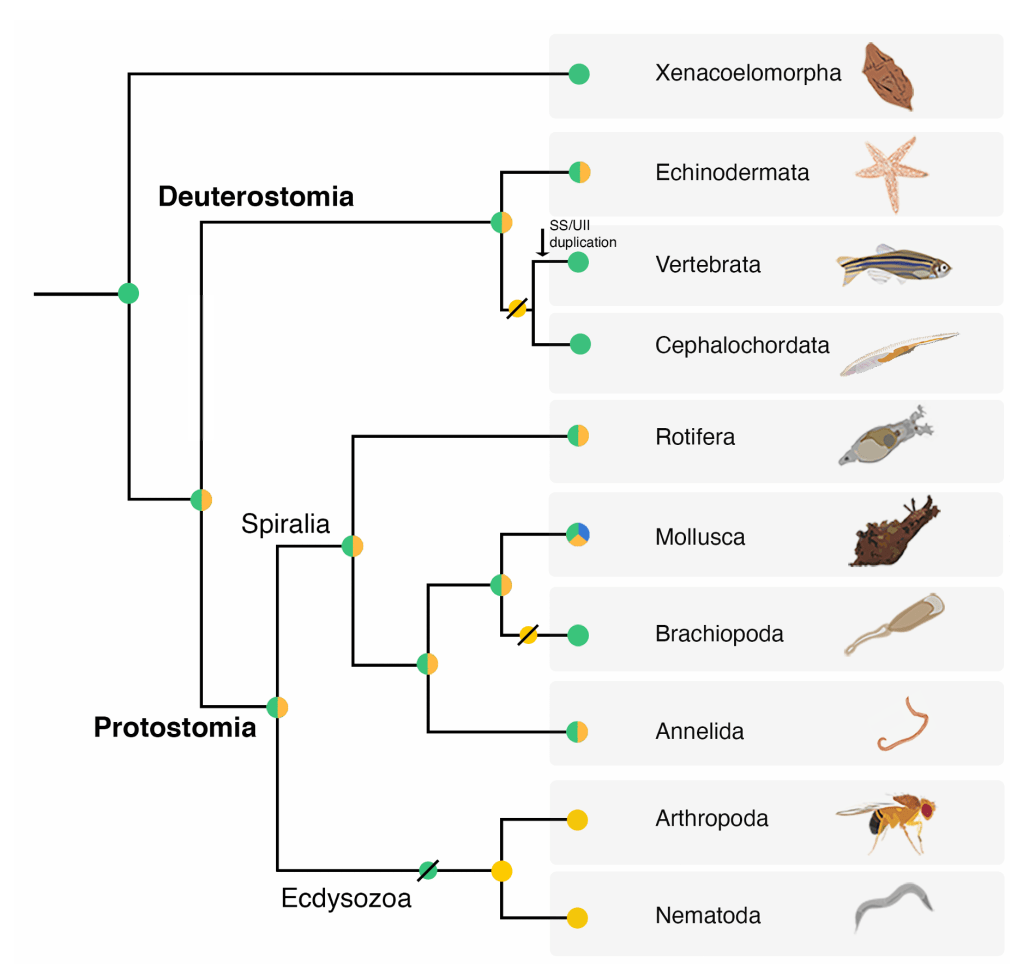
Our group is particularly interested in a subset of toxins that mimic endogenous signaling molecules (peptide hormones and neuropeptides) in prey. These molecules have the potential to not only inform on the design of improved drug leads for disease but can also be used as tools to elucidate the molecular evolution of toxins and the hormones they mimic. In the past we showed that cone snail insulins were recruited into venom where they expanded and diversified in response to prey taxa (e.g., fish-hunting cone snail insulins evolved to target the fish insulin receptor while insulins from snail hunters mimic insulins expressed in snails. More recently, we showed that venom somatostatins also evolved from an endogenous somatostatin-like signaling gene and, upon recruitment into venom, diversified into a large family of somatostatin-like toxins (> 400 sequences). In this most recent study, we further showed that, as observed for insulins, venom somatostatins mimic the hormone expressed in prey and elucidated a previously unknown somatostatin-like signaling system in invertebrates. Based on these findings we postulate that somatostatin and its receptor evolved more than 600 million years ago making it one of the oldest signaling systems in animals.
Publication on the evolution of (venom) somatostatins; Publication on the evolution of venom insulins

Biosynthetic Enzymes
At any given time, hundreds of highly modified, structurally complex peptides are biosynthesized in the venom gland of cone snails. We have had a long-standing interest in identifying these enzymes and utilizing evolutionary and functional information of these enzymes to generate optimized venom expression systems. Together with Prof. Ellgaard at the University of Copenhagen we have commenced to successfully express many of the new toxins we have recently discovered, including venom insulins and other complex peptides with multiple disulfide bonds.